
OPUS LAP 2021
CO2CHEM - REDOX-NEUTRAL PHOTOCATALYTIC C-H CARBOXYLATION OF HYDROCARBONS WITH CO2
Biological photosynthesis is the essential molecular process for life on earth converting solar energy into energy-enriched molecules, such as ATP or carbohydrates. From this perspective, the products of biological photosynthesis are either used as molecular batteries or as building blocks for the biochemical synthesis of plant material. Recent years have seen large efforts in mimicking biological photosynthesis by technical processes with the goal of converting solar energy into chemically stored energy. Such artificial photosyntheses, e.g., combining photovoltaic charge separation and water electrolysis, using hybrid biotechnological systems or semi-conductor photocatalysis have already reached high technological levels up to pilot plants. The energy-rich molecules produced in such processes are typically dihydrogen, methane or methanol from the reduction of carbon dioxide or syngas (H2 / CO) as feed for the Fischer-Tropsch process. A recent study summarized the state of the art and the currently pursued Kopernikus projects in Germany promise progress in the provision of solar fuels. In contrast, the solar driven synthesis of valuable chemicals using carbon dioxide is still at a very early stage of development.
So far, carbon dioxide is only used to a small extend as raw material in chemical synthesis, but industrial projects in polymer production have shown the feasibility and sustainability of the approach. However, a chemical activation of carbon dioxide in thermal reactions requires reactive reaction partners. Solar driven reactions may therefore provide advantageous alternatives.
This proposal aims to develop the synthetic and mechanistic basis for the photocatalytic, redox-neutral C-H carboxylation of saturated hydrocarbons with carbon dioxide (Scheme 1).
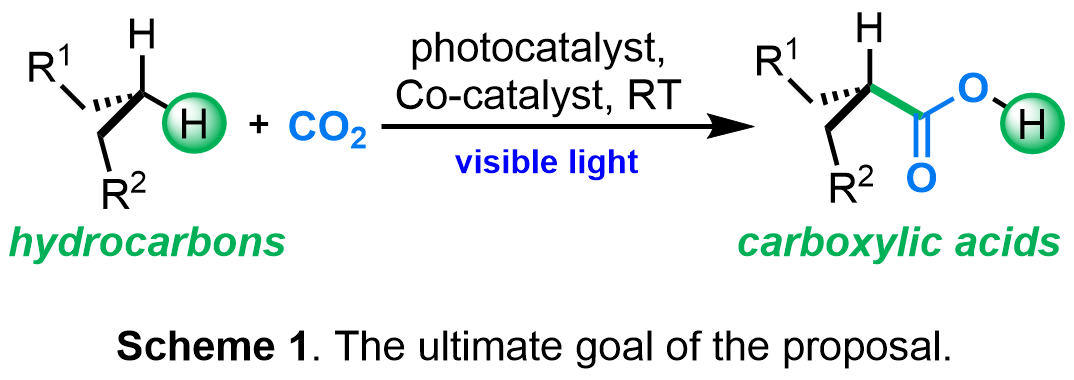 Our preliminary data clearly show that CO2 is a valuable reagent in functionalizations of C-H bonds in which carbon dioxide is formally inserted into a carbon hydrogen bond. In particular, the König group, realized the redox neutral carboxylation of arenes, styrenes as well as benzylic or α-carbonyl C-H bonds (Scheme 2). The developed methods are highly valuable and proved useful in the synthesis of drug molecules, such as ibuprofen, from simple arene precursors and carbon dioxide. It should be clearly stated, however, that both existing methods preclude the use of the most abundant, non activated C(sp3)-H bonds.
Our preliminary data clearly show that CO2 is a valuable reagent in functionalizations of C-H bonds in which carbon dioxide is formally inserted into a carbon hydrogen bond. In particular, the König group, realized the redox neutral carboxylation of arenes, styrenes as well as benzylic or α-carbonyl C-H bonds (Scheme 2). The developed methods are highly valuable and proved useful in the synthesis of drug molecules, such as ibuprofen, from simple arene precursors and carbon dioxide. It should be clearly stated, however, that both existing methods preclude the use of the most abundant, non activated C(sp3)-H bonds.
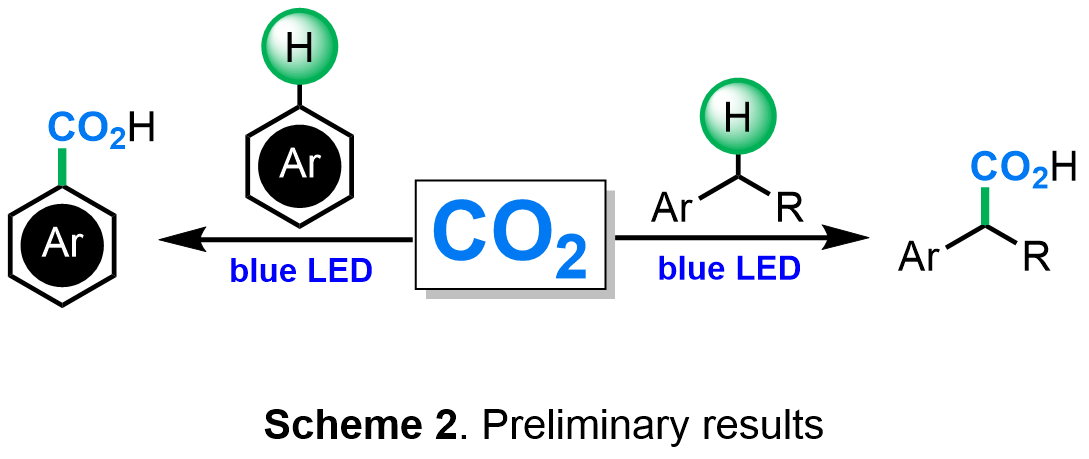
In order to address this challenge, we propose new synthetic strategies based on photocatalytically generated alkyl radicals and earth-abundant metal complexes to provide C-H carboxylation in an overall redox-neutral process.
Nature optimized the photosynthetic mechanisms over millions of years by evolution. In contrast, humankind is in a hurry and immediate advances are needed. Though we are aware that even the extensive use of CO2 for chemical production will not solve the global carbon emission problem, the envisioned carboxylation of hydrocarbons’ will utilize anthropogenic carbon dioxide and as such contribute to the reduction of carbon footprint.


