Vinyloxy-alkoxides: A new Aldol PRINCIPLE
Hartmut Redlich
Organisch-Chemisches Institut, Westfälische Wilhelms-Universität,
Corrensstraße 40, 48149 Münster, Germany
Email: redlich@uni-muenster.de
The reaction of 2-bromomethyl-1,3-dioxolane 1 with magnesium followed by reaction with carbonyl functions provides surprisingly the same coupling products as the reaction of 2-vinyloxy-ethanol 2 with ethylmagnesium bromide (see scheme)[1,2,3].
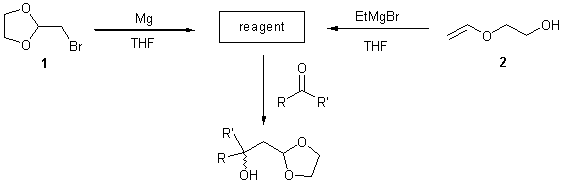
The stereochemical behavior of this new type of aldol reaction can be easily controlled. It offers most excellent yields and high stereoselectivities for diastereomers and enantiomers. The stereochemical information can be introduced either by the reagent (substituted at the olefinic part or in the dioxolane moiety), the reactant, the counterion or any combination of these (see scheme).
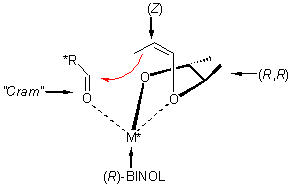
The application of vinyloxy-alkoxides on carbohydrate derivatives requires carbo-hydrate vinyl ethers with an unprotected hydroxy group close to the vinyl ether moiety. These reagents exhibit a pronounced carbon-carbon-coupling activity towards carbonyl functions.
References
[1] H. Ida, N. Yamazaki, C. Kibayashi, J. Org. Chem. 1986, 51, 4245-4249.
[2] M. Schmeichel, H. Redlich, Synthesis 1996, 1002-1006.
[3] P. Maier, H. Redlich, Synlett 2000, 257-259.