sulfoneS in glycochemistry :
scouting for new tracks
Patrick Rollin
ICOA, UMR 6005, Université d’Orléans, BP 6759, F-45067 Orléans, France
patrick.rollin@univ-orleans.fr
Selective introduction-expulsion of protective groups still remains a major concern in carbohydrate synthetic strategies and cyclic acetal systems have proven to be particularly effective in that respect.
As part of ongoing projects centered on diversely thiofunctionalized sugars, we have developed an original method for the temporary protection of saccharides by b-sulfonylated cyclic acetals, readily obtainable under alkaline conditions using commercially available Z- and E-1,2-bis(phenylsulfonyl)ethylenes (BPSE)1,2 :
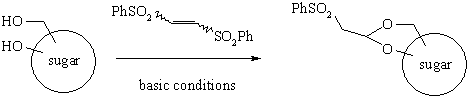
Following a double Michael addition pathway, BPSE readily combined with 1,2- and 1,3-diols or polyols in various carbohydrate series to afford in preparative yields (70-90%) phenylsulfonylethylidene (PSE) acetals.
A thorough examination of ring-opening ability of the preceding acetals is being performed in our laboratory. They resist most Lewis acids3 and they show a remarkable reluctance to acid-catalyzed hydrolysis : in particular conditions (TFA 90% at 60°C), benzyl ethers can themselves undergo hydrolytic cleavage when PSE acetals remain in place.
In contrast, LiAlH4 easily brings about expulsion of the PSE clip in anhydrous medium : however, the strong reducing character of the reagent precludes a routine use for the deprotection of PSE-equipped saccharides. Investigation towards milder conditions has shown that standard ethanolysis in basic medium - 1M KOH or even better, alkaline carbonates – allows comparable results, which characterise in fact a quite unusual transacetalation under alkaline conditions.
Further results4,5 will be presented to illustrate the synthetic potential of the sulfone functional group in glycochemistry.
[1] Chéry, F. ; Rollin, P. ; De Lucchi, O. ; Cossu, S. Synthesis, 2001, 286-292
[2] Cabianca, E. ; Chéry, F. ; Rollin, P. ; Tatibouët, A. ; De Lucchi, O. Tetrahedron Lett. 2002, 43, 585-587
[3] Chevalier-du Roizel, B. ; Cabianca, E. ; Rollin, P. ; Sinaÿ, P. Tetrahedron 2002, 58, 9579-9583
[4] Chéry, F. ; Desroses, M. ; Tatibouët, A. ; De Lucchi, O. ; Rollin, P. Tetrahedron, 2003, 59, 4563-4572.
[5] Cabianca, E. ; Chéry, F. ; Pillard, C. ; Tatibouët, A. ; De Lucchi, O. ; Rollin, P. Tetrahedron Lett., 2003, 44, 5723-5725.